Une nouvelle publication dans GLIA pour P3M

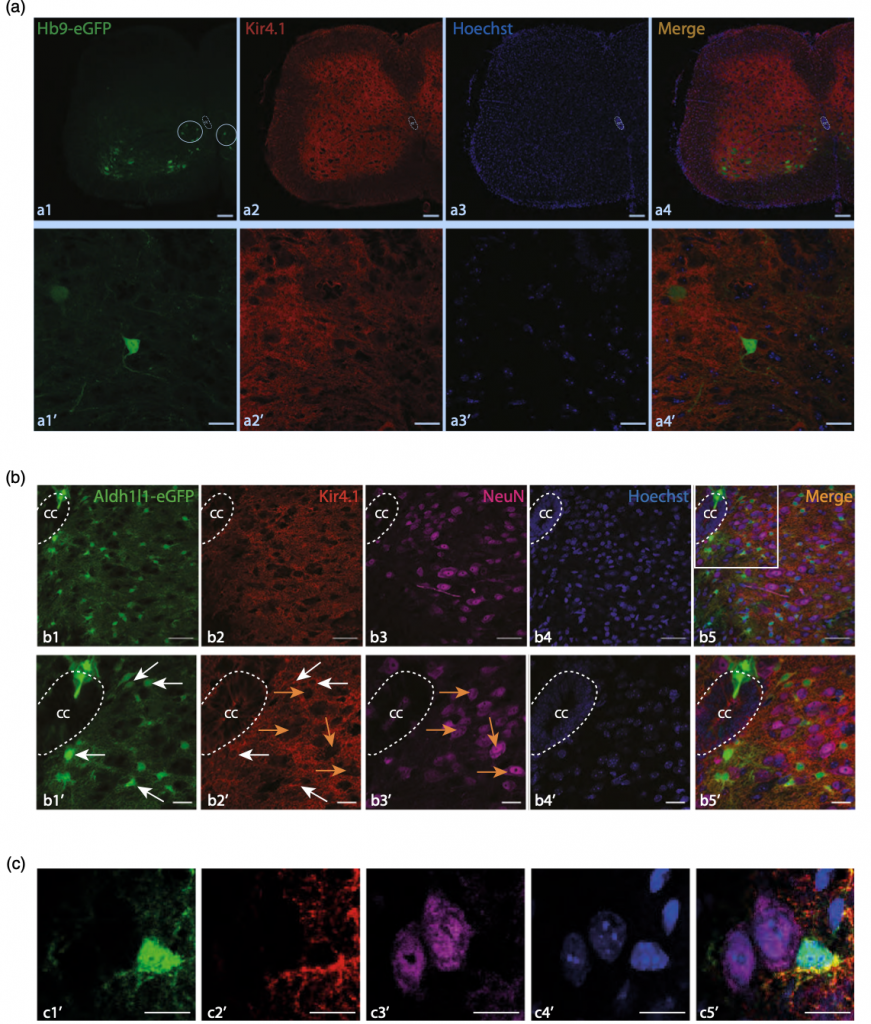
“Astrocytic Kir4.1 channels regulate locomotion by orchestrating neuronal rhythmicity in the spinal network”
Barbay T., Pecchi E., Ducrocq M., Rouach N., Brocard F., Bos R.
http://doi.org/10.1002/glia.24337
Abstract:
Neuronal rhythmogenesis in the spinal cord is correlated with variations in extracellular K+ levels ([K+]e). Astrocytes play important role in [K+]e homeostasis and compute neuronal information. Yet it is unclear how neuronal oscillations are regulated by astrocytic K+ homeostasis. Here we identify the astrocytic inward-rectifying K+ channel Kir4.1 (a.k.a. Kcnj10) as a key molecular player for neuronal rhythmicity in the spinal central pattern generator (CPG). By combining two-photon calcium imaging with electrophysiology, immunohistochemistry and genetic tools, we report that astrocytes display Ca2+ transients before and during oscillations of neighbouring neurons. Inhibition of astrocytic Ca2+ transients with BAPTA decreases the barium-sensitive Kir4.1 current responsible of K+ clearance. Finally, we show in mice that Kir4.1 knockdown in astrocytes progressively prevents neuronal oscillations and alters the locomotor pattern resulting in lower motor performances in challenging tasks. These data identify astroglial Kir4.1 channels as key regulators of neuronal rhythmogenesis in the CPG driving locomotion.
Significance Statement:
Despite decades of research, the cellular mechanisms responsible for the synchronized rhythmic oscillations driving locomotion remain elusive. To gain insight into the function of the spinal locomotor network, numerous studies have characterized diverse classes of locomotor-related neurons to determine their role in generating rhythmic movements during locomotion. In contrast, studies investigating non-neuronal components of the spinal cord are sparse. Our study represents a significant breakthrough by identifying astrocytic K+ uptake as a key regulator of neuronal rhythmicity synchronization and locomotor pattern at the cellular, microcircuit and system levels. These data provide mechanistic insights into the neuroglial dialogue at play during rhythmogenesis and point to a novel astroglial target for restoring normal neuronal network excitability in brain disorders and neurodegenerative diseases.